And since we're doing formal charge, we need to think about all I could put a four here, and from that four we're going to subtract the number of valenceĮlectrons in the bonded atom or the number of valence electrons that carbon has around it in our drawing. We already know thatĬarbon is supposed to have four valence electrons, so Number of valence electrons that carbon is supposed to have. Number of valence electrons in the free atom or the So if we want to assign aįormal charge to carbon, we need to think about the Same for the bond betweenĬarbon and hydrogen, right? Each bond consists of two electrons, so I can go around and put inĪll of my bonding electrons. So the bond between oxygen and carbon consists of two electrons. The bonding electrons or the electrons in thoseīonds around carbon, and we know that each bondĬonsists of two electrons. 
Structure on the left here, which is a dot structure for methanol, and let's assign aįormal charge to carbon. So one definition for formal charge is the hypothetical charge that would result if all bonding electronsĪre shared equally. Both formal charge and oxidation states are ways of counting electrons, and they're both very useful concepts. Regardless of what system we use, the carbon is still satisfied because it has an octet of electrons. Both of these system simply account for electrons to make the math simpler. 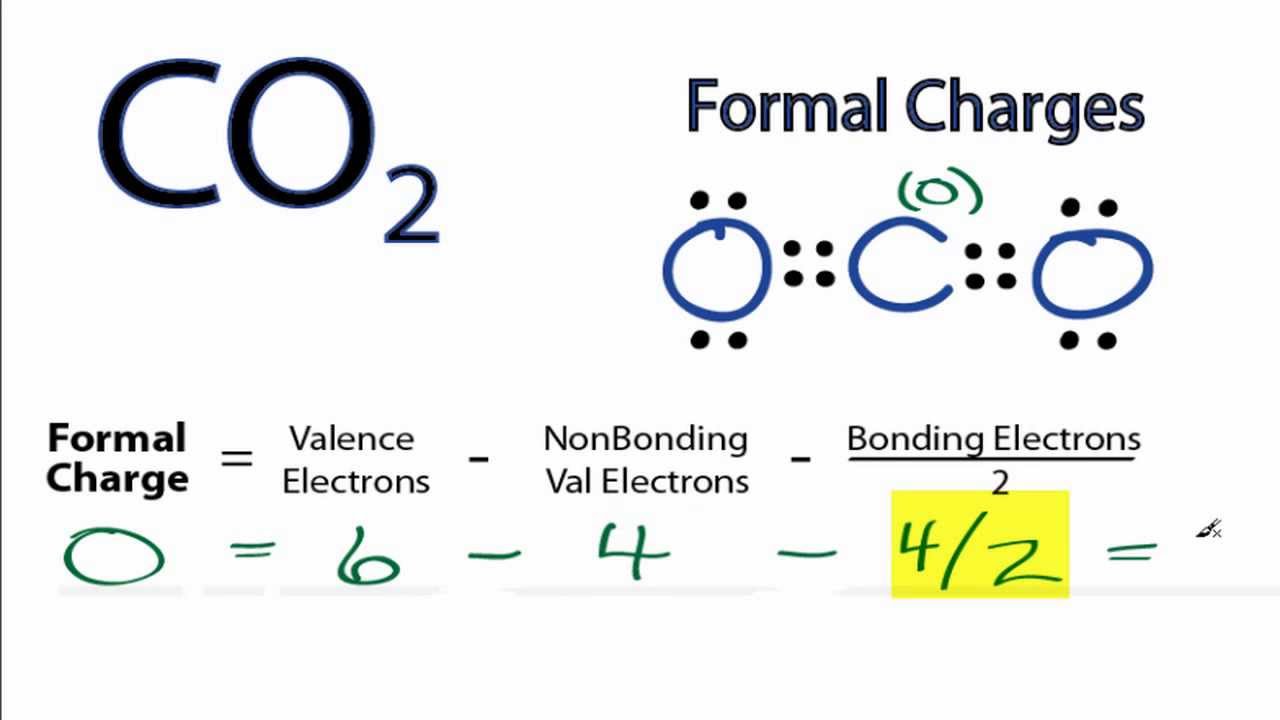
The only difference between the two is that formal charge doesn’t take into account electronegativity, while oxidation number does. For a molecule to be polar it requires polar bonds and certain molecular geometries so the dipole moments of the polar bonds don’t cancel.įormal charge and oxidation number are just convenient ways of assigning electrons to atoms in molecules. Entire molecules are most often where we use polarity and it describes a molecule with a negative and positive pole (or side). Individual bonds can be polar if the bonding atoms have a high enough electronegativity difference, and therefore the more electronegative atom pulls much stronger on the electrons than the other. In a covalent bond, the bonding electrons spend more time around the more electronegative atom as opposed to the less electronegative one.īeing polar in this context means one end has an excess of negative charge while the other has a lack of negative charge (or is positive). Electronegativity is the measure of this attraction elements have for electrons. Certain elements have more attraction to electrons than others and so the electrons end up spending more time around one atom compared to the other in a bond. It’s sharing those eight electrons with the hydrogen and oxygen atoms in covalent bonds, but the sharing isn’t equitable. It has an octet (8) of electrons from bonding with the hydrogen and oxygen atoms. (a) The 78.86-carat Ahmadabad diamond, a historic Indian gem purchased in Gujarat in the 17th century by the French explorer Jean-Baptiste Tavernier and sold in 1995 for $4.3 million, is a rare example of a large single crystal of diamond, the less-stable allotrope of carbon.Well the carbon is satisfied with the number of electrons it has in methanol. \): Crystalline Samples of Carbon and Silicon, the Lightest Group 14 Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
